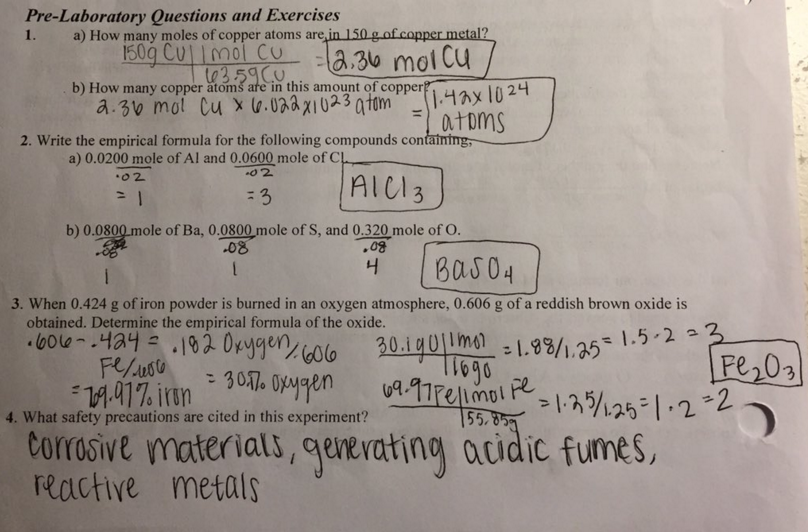Empirical Formula Lab
Observations:
1. When we added the HCl to the Mg, if began to fizz and bubble, releasing a gas as seen in the video
2. The Mg strips were about .5in-1in. long each.
3. The Mg strips slowly but surely dissolved, and the solution looked clearish/whitish
4. As we heated and cooled the dish, there seemed to be less and less of the solution, and it looked even more white.
1. When we added the HCl to the Mg, if began to fizz and bubble, releasing a gas as seen in the video
2. The Mg strips were about .5in-1in. long each.
3. The Mg strips slowly but surely dissolved, and the solution looked clearish/whitish
4. As we heated and cooled the dish, there seemed to be less and less of the solution, and it looked even more white.
Pre-Lab Questions
Errors/Calculations:
There were two very possible errors in our experiment. As seen before, the mass of the Cl was negative, which means we lost some. How is this possible? Well, we believe either spattering occurred or that our dish got switched with another teams after the 4th reading of mass. As our dish was being heated, our solution could have got splashed out of the dish from the boiling. This would cause a reading of less product than we started with. The other situation is that another team took our dish, since there was such a massive and unexpected decrease in mass. If this happened, then another groups data would also be off. To prevent this we have to get our labs done quickly instead of doing it over the course of a week, and keep a closer eye on our dish.
1.Mass of evaporating dish-
47.39g
2. Mass of dish and Mg-
47.87g
3. Mass of Mg:
.49g
4. Mass of dish & MgCl each time it's heated and cooled-
50.98g, 48.37g, 48.31g, 48.3g, 47.66g, 47.66g
5. Mass of MgCl2-
.28g
6. Mass of Cl in MgCl2 -
.21g (actually negative, but for the sake of calculations we will keep it positive)
7. Moles of Mg-
.49gMg x 1 mol Mg
24.3gMg
= .0202 mol Mg
8. Moles of Chlorine-
.21gCl x 1 molCl
35.5gCl
= .0059 mol Cl
9. Moles of Mg/ Smallest number of moles-
3.4
10. Moles of Cl/ Smallest number of moles-
1
11. Your experimental empirical formula-
(x5 for whole numbers)-- Mg17Cl5
12. True empirical formula-
MgCl2
13. Percent Error-
.28/1.92= 1.64 -> 1.64/1.92= .8542 x 100 =
85.42% (yikes)
14. Balanced equation-
Mg + 2HCl-->MgCl2 + H2
There were two very possible errors in our experiment. As seen before, the mass of the Cl was negative, which means we lost some. How is this possible? Well, we believe either spattering occurred or that our dish got switched with another teams after the 4th reading of mass. As our dish was being heated, our solution could have got splashed out of the dish from the boiling. This would cause a reading of less product than we started with. The other situation is that another team took our dish, since there was such a massive and unexpected decrease in mass. If this happened, then another groups data would also be off. To prevent this we have to get our labs done quickly instead of doing it over the course of a week, and keep a closer eye on our dish.
1.Mass of evaporating dish-
47.39g
2. Mass of dish and Mg-
47.87g
3. Mass of Mg:
.49g
4. Mass of dish & MgCl each time it's heated and cooled-
50.98g, 48.37g, 48.31g, 48.3g, 47.66g, 47.66g
5. Mass of MgCl2-
.28g
6. Mass of Cl in MgCl2 -
.21g (actually negative, but for the sake of calculations we will keep it positive)
7. Moles of Mg-
.49gMg x 1 mol Mg
24.3gMg
= .0202 mol Mg
8. Moles of Chlorine-
.21gCl x 1 molCl
35.5gCl
= .0059 mol Cl
9. Moles of Mg/ Smallest number of moles-
3.4
10. Moles of Cl/ Smallest number of moles-
1
11. Your experimental empirical formula-
(x5 for whole numbers)-- Mg17Cl5
12. True empirical formula-
MgCl2
13. Percent Error-
.28/1.92= 1.64 -> 1.64/1.92= .8542 x 100 =
85.42% (yikes)
14. Balanced equation-
Mg + 2HCl-->MgCl2 + H2
Post-Lab Questions