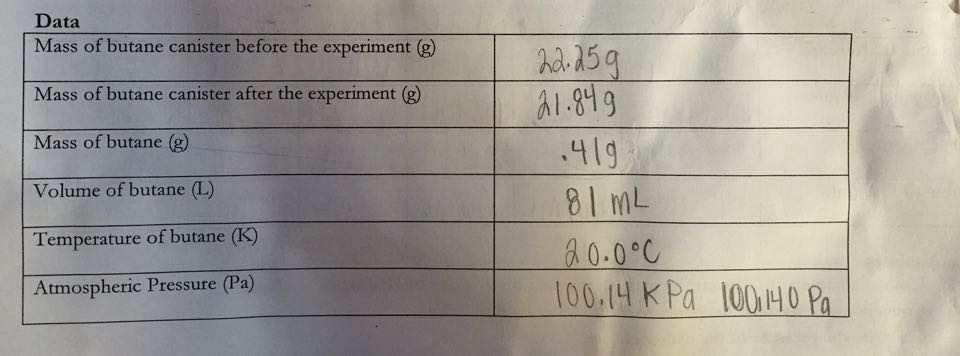Gas Law Lab 11/6/15 AP Chem
Determining the Gas Constant and Molecular Mass of Butane
Problem Statement:
IV:
Part One: Adding Mg to HCl
Part Two: Adding butane
DV:
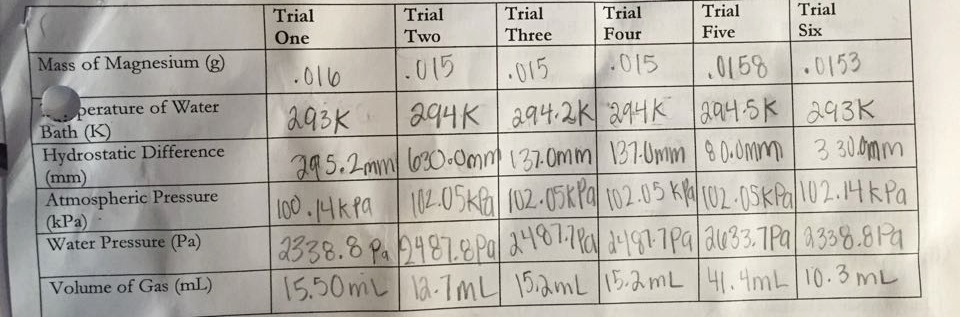
Part One: volume of gas and hydrostatic difference
Part Two: experimental mass of one mole of butane
Control:
Part One: Atmosphere pressure (used to find R value)
Part Two: Chemical formula of C4H10
Constants:
Part One: Amount of Mg, Amount of HCl, and thermometer
Part Two: Butane canister, thermometer, trough, flask
Purpose:
Part One: Determine the Gas Constant (R) using magnesium and hydrochloric acid.
Part Two: Determine the molecular mass of butane experimentally
Hypothesis:
Part One: If we add Mg to HCl, then we will find a correct R value, because a reaction will occur and hydrogen gas will be produced, giving us the components to correctly calculate the gas constant.
Part Two: If we use standard conditions and release butane into the flask correctly, then we will get the mass per mole close to the chemical findings- 58.14g/mol- because the different components of this experiment (mass, volume, and temp of butane, and atm pressure), will allow us to correctly calculate the molecular mass.
Materials:
Part One: Mg, HCl, Beaker of water, clamp, thermometer, stopcock, buret, tub, running water, ruler
Part Two: Trough, water, butane canister, flask, graduated cylinder, marker, thermometer
Procedure:
Part One:
IV:
Part One: Adding Mg to HCl
Part Two: Adding butane
DV:
Part One: volume of gas and hydrostatic difference
Part Two: experimental mass of one mole of butane
Control:
Part One: Atmosphere pressure (used to find R value)
Part Two: Chemical formula of C4H10
Constants:
Part One: Amount of Mg, Amount of HCl, and thermometer
Part Two: Butane canister, thermometer, trough, flask
Purpose:
Part One: Determine the Gas Constant (R) using magnesium and hydrochloric acid.
Part Two: Determine the molecular mass of butane experimentally
Hypothesis:
Part One: If we add Mg to HCl, then we will find a correct R value, because a reaction will occur and hydrogen gas will be produced, giving us the components to correctly calculate the gas constant.
Part Two: If we use standard conditions and release butane into the flask correctly, then we will get the mass per mole close to the chemical findings- 58.14g/mol- because the different components of this experiment (mass, volume, and temp of butane, and atm pressure), will allow us to correctly calculate the molecular mass.
Materials:
Part One: Mg, HCl, Beaker of water, clamp, thermometer, stopcock, buret, tub, running water, ruler
Part Two: Trough, water, butane canister, flask, graduated cylinder, marker, thermometer
Procedure:
Part One:
Part Two:
Data:
Part One:
Part One:
Part Two:
Pre-Lab Questions:
Post-Lab Questions: Still waiting on 5 R-values to compare for this section
Calculations:
Conclusion:
Still waiting on 5 R-values to compare for this section
Discussion/ Reflection:
Still waiting on 5 R-values to compare for this section