Acid in Juice Titration - AP Chem pd.4 Rileigh Robertson
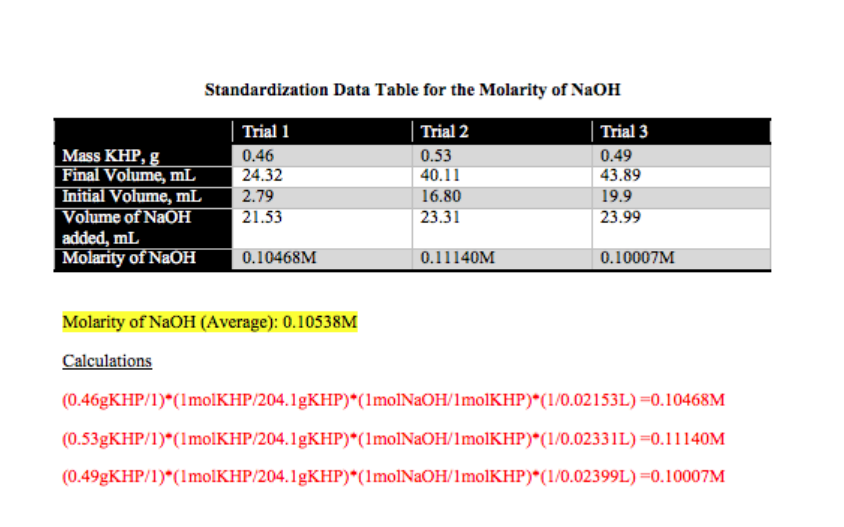
Standardization of NaOH
Pre-lab Questions:
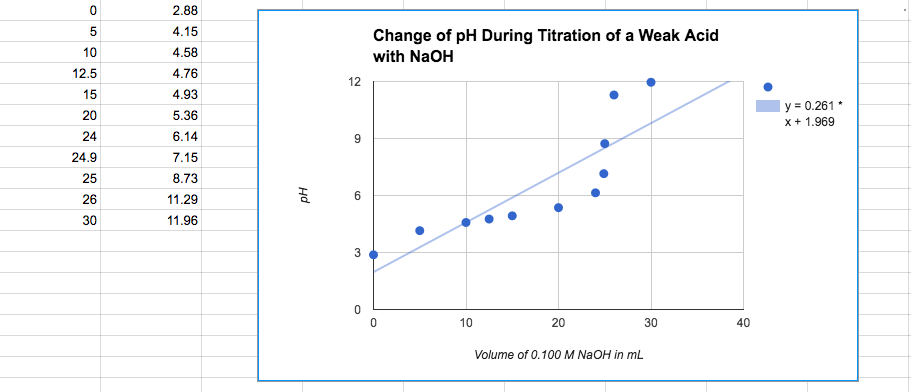
Graph for 7 below
b) ph=7.15
d) I would use bromthymol blue as my indicator because the the equivalence point is in the range for color change.
b) ph=7.15
d) I would use bromthymol blue as my indicator because the the equivalence point is in the range for color change.
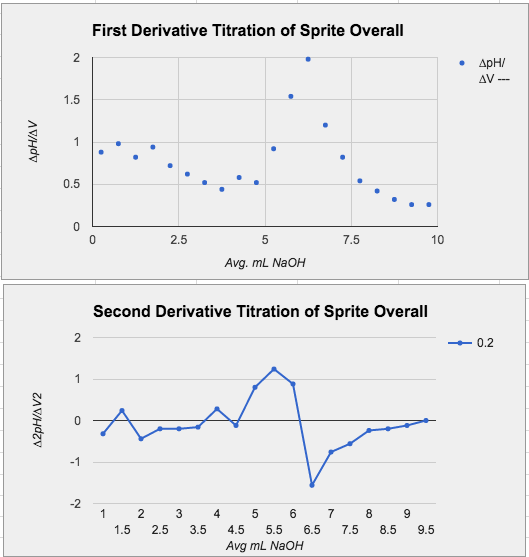
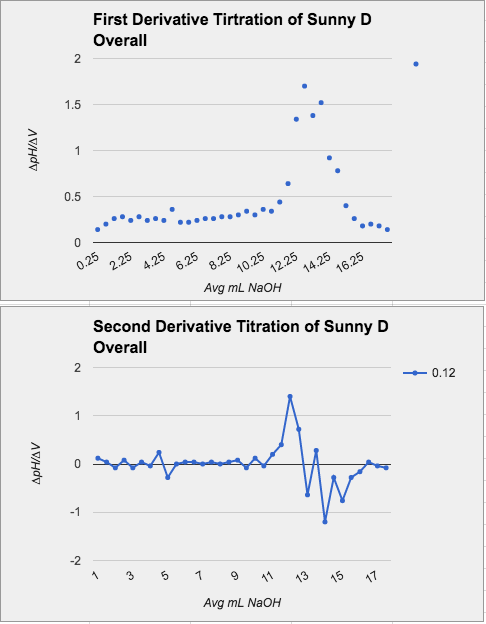
Data Tables and Graphs for Sunny D and Sprite
Note: We didn't use an indicator, we used a pH sensor measure the point where equivalence occurs.
Argumentation: If I were to do this experiment again, I would want to use an indicator in addition to the pH sensor just for extra assurance.
Post-Lab Questions:
1. The acid concentration may be less or more than what it actually is because since beaker or graduated cylinder measurements are not precise enough, when you fill up the solution to the line it could be slightly less or more than the actual volume. This would make the concentration of the acid either a little more or a little less than what it actually is. It affects my number of significant figures because there would be less significant figures, which makes my answer, in this case, the concentration of acid, less precise.
2. The concentration of acid would be less because the water will dilute the acid content in the solution, effecting the products as well.
3. Groups that titrated a dark soda had to take the step of using a pH sensor in their experiment to tell when the indicator changed colors. Since the soda is dark, you wouldn't be able to see the indicator change color at the equivalence point, so a pH sensor would be used in the beaker as well, which would display the changing pH of the solution.
4. THe average calculated acid concentration would be less than it was supposed to be because adding more base than what it was at the equivalence point would push it past equivalence and make it more concentrated of the base.
5.
6.
7. I would tell them to neutralize their acidic snacks or drinks with a base that could be concentrated in certain foods.
1. The acid concentration may be less or more than what it actually is because since beaker or graduated cylinder measurements are not precise enough, when you fill up the solution to the line it could be slightly less or more than the actual volume. This would make the concentration of the acid either a little more or a little less than what it actually is. It affects my number of significant figures because there would be less significant figures, which makes my answer, in this case, the concentration of acid, less precise.
2. The concentration of acid would be less because the water will dilute the acid content in the solution, effecting the products as well.
3. Groups that titrated a dark soda had to take the step of using a pH sensor in their experiment to tell when the indicator changed colors. Since the soda is dark, you wouldn't be able to see the indicator change color at the equivalence point, so a pH sensor would be used in the beaker as well, which would display the changing pH of the solution.
4. THe average calculated acid concentration would be less than it was supposed to be because adding more base than what it was at the equivalence point would push it past equivalence and make it more concentrated of the base.
5.
6.
7. I would tell them to neutralize their acidic snacks or drinks with a base that could be concentrated in certain foods.
Planning Sheet
Background: The acids in Sprite are carbonic acid and citric acid, with their Ka (respectively) being 4.3*10^-7 (pH=6.4) and 8.4*10^-4 (pH=3.1). The acids in Sunny D are ascorbic acid and citric acid, with their Ka (respectively) being 7.9*10^-5 (pH=4.1) and 8.4*10^-4 (pH=3.1). We chose the indicator based on the acid with the lowest pH, which would be citric acid. However, we do not have an indicator that changes color that low, so we have to use a universal indicator.
Experimental Design
Title: Acid in Juice Titration
Purpose: To figure out how much acid is in Sprite and Sunny D.
Prediction: I believe that, for both drinks, the acid makes up a very little portion of the whole amount of substance in the bottles.
Independent Variable: Volume of NaOH added to both drinks
Dependent Variable: pH of the solution
Trials: 5
Constants: amount of NaOH added to Sprite, amount of NaOH added to Sunny D, pH probe, temperature of each drink, amount of Sprite and Sunny D titrated, the indicator used, same buret, number of trials for each drink
Control Setup: Titrating NaOH with KHP
Testable Research Question: How much acid is in Sunny D and Sprite?
Hypothesis: If there is acid in Sunny D and Sprite, then the concentration would not be very big because there are a lot more of other things in each drink than there is acid.
Materials list: buret, ring stand, ring stand clamp, 100 mL each of Sunny D and Sprite, ten 50 mL beakers, pH probe, device to read the pH from the probe (LabQuest), two 10 mL pipettes, two 200 mL beakers, magnetic stirrer, universal indicator, disposable pipette, 140 mL of 0.1 NaOH solution, a buffer solution that is a pH of 4, a buffer solution at a pH of 10, tape
Procedure List:
Background: The acids in Sprite are carbonic acid and citric acid, with their Ka (respectively) being 4.3*10^-7 (pH=6.4) and 8.4*10^-4 (pH=3.1). The acids in Sunny D are ascorbic acid and citric acid, with their Ka (respectively) being 7.9*10^-5 (pH=4.1) and 8.4*10^-4 (pH=3.1). We chose the indicator based on the acid with the lowest pH, which would be citric acid. However, we do not have an indicator that changes color that low, so we have to use a universal indicator.
Experimental Design
Title: Acid in Juice Titration
Purpose: To figure out how much acid is in Sprite and Sunny D.
Prediction: I believe that, for both drinks, the acid makes up a very little portion of the whole amount of substance in the bottles.
Independent Variable: Volume of NaOH added to both drinks
Dependent Variable: pH of the solution
Trials: 5
Constants: amount of NaOH added to Sprite, amount of NaOH added to Sunny D, pH probe, temperature of each drink, amount of Sprite and Sunny D titrated, the indicator used, same buret, number of trials for each drink
Control Setup: Titrating NaOH with KHP
Testable Research Question: How much acid is in Sunny D and Sprite?
Hypothesis: If there is acid in Sunny D and Sprite, then the concentration would not be very big because there are a lot more of other things in each drink than there is acid.
Materials list: buret, ring stand, ring stand clamp, 100 mL each of Sunny D and Sprite, ten 50 mL beakers, pH probe, device to read the pH from the probe (LabQuest), two 10 mL pipettes, two 200 mL beakers, magnetic stirrer, universal indicator, disposable pipette, 140 mL of 0.1 NaOH solution, a buffer solution that is a pH of 4, a buffer solution at a pH of 10, tape
Procedure List:
- Rinse your buret with distilled water 3 times and then with a little NaOH 3 times
- Set up your buret with the ring stand and the ringstand clamp and fill the buret to the 0mL mark
- Calibrate your pH probe with the 2 buffer solutions (put the probe in the solution with the lower pH first)
- Fill one of the 200 mL beakers with about 120 mL of Sprite
- Use one of the 10 mL pipettes to transfer 20mL of Sprite to each of five 50mL beakers
- Place one beaker onto the magnetic stirrer with the little magnetic in the beaker and turn it on
- Make sure the buret is over the beaker
- Once the pH probe is done calibrating, place it in the beaker and tape it to the buret so you do not have to hold it
- Place 3 drops of the indicator into the Sprite
- Record the starting pH in a data table
- This is where you begin your titration: open the stopcock to allow 0.5mL of NaOH to fall into the solution
- Close the stopcock and record the pH
- Do steps 11 and 12 until you have reached the point where 10 mL of NaOH has been added
- Repeat steps 6 through 13 with the remaining beakers of Sprite
- Repeat steps 4 through 14 with the Sunny D, omitting step 9 and titrating with 18mL of NaOH instead of 10 (you will have to refill the buret to complete all 5 trials)
- Clean up once all the titrations have been completed
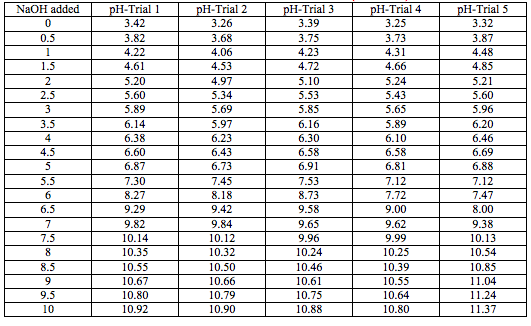
Data Tables:
Titration of Sprite
Reactions: 2NaOH+H2CO3 --> Na2CO3+H2O (carbonic acid)
3NaOH+H3C6H5O7 --> 3H2O+Na3C6H5O7 (citric acid)
Reactions: 2NaOH+H2CO3 --> Na2CO3+H2O (carbonic acid)
3NaOH+H3C6H5O7 --> 3H2O+Na3C6H5O7 (citric acid)
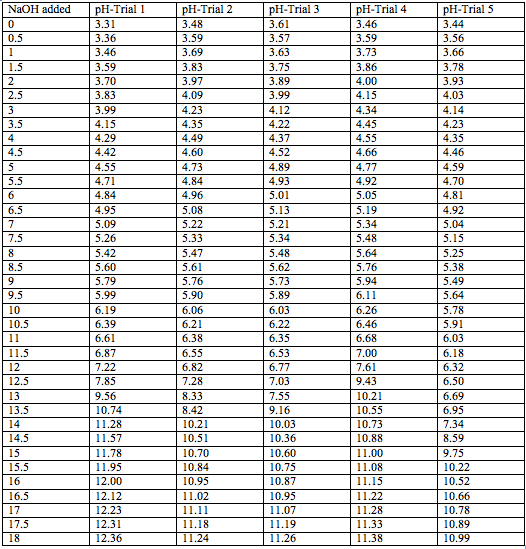
Sunny D