Redox Titration of Iron and Oxalic Acid 9/14/15
Problem Statement: The purpose of this lab is to standardize a solution of potassium permanganate by redox titration with a standard solution of iron (II) ions. Then, a solution of oxalic acid is then titrated with the permanganate solution to determine the exact concentration of oxalic acid.
Hypothesis: If we find the molarity of the potassium permanganate after it is standardized, then we can find the concentration of oxalic acid, because using a mole to mole conversion of the permanganate and the moles of oxalic acid found from the data would produce the molarity if set above the volume.
Procedure:
Independent variable: Potassium permanganate
Dependent variable: Concentration of the permanganate after standardization, concentration of oxalic acid after titrated
Constants: Distilled water, amount of iron (II) put in for each trial, amount of oxalic acid put in for each trial, temp warmed to on the hot plate
Control: .1 M Fe2+
Independent variable: Potassium permanganate
Dependent variable: Concentration of the permanganate after standardization, concentration of oxalic acid after titrated
Constants: Distilled water, amount of iron (II) put in for each trial, amount of oxalic acid put in for each trial, temp warmed to on the hot plate
Control: .1 M Fe2+
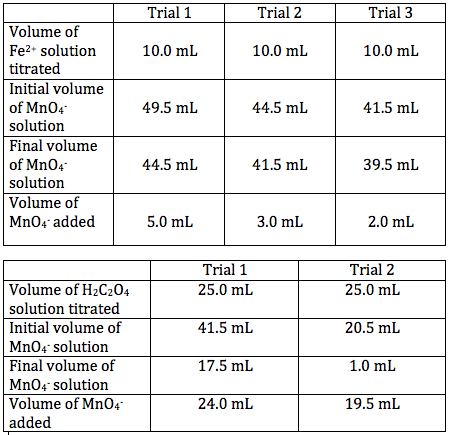
Data Tables for the Titrations:
Analysis:
Postlab questions:
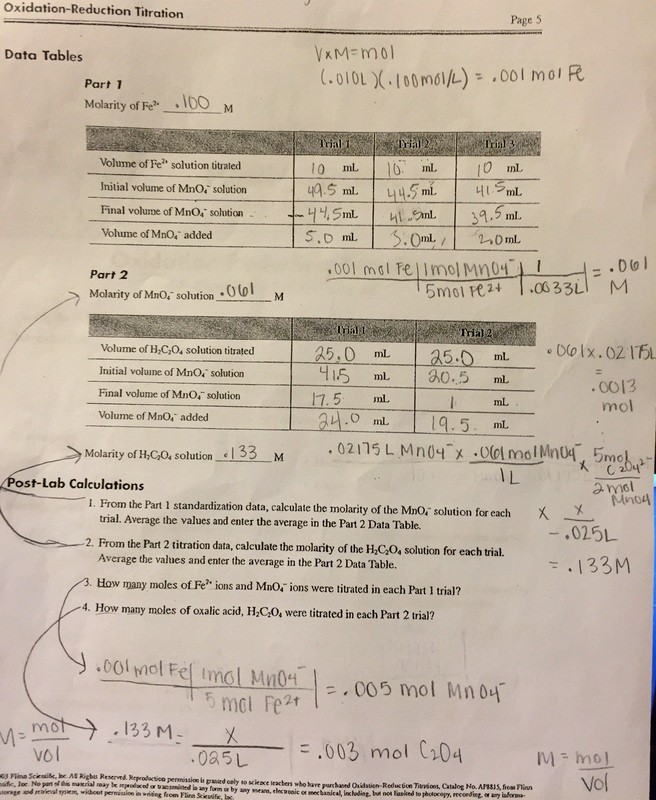
1) .061 M
2) .133 M
3) .005 mol MnO4-
4) .003 mon C2O4
work: (sorry for the mess)
Postlab questions:
1) .061 M
2) .133 M
3) .005 mol MnO4-
4) .003 mon C2O4
work: (sorry for the mess)
Conclusion:
Throughout the experiment, my hypothesis stood true. I started with moles of iron, followed all of the procedure, and did calculations to finish with the concentration of oxalic acid as said in my hypothesis. I stated that if we find the molarity of the potassium permanganate after it is standardized, then we can find the concentration of oxalic acid, because using a mole to mole conversion of the permanganate and the moles of oxalic acid found from the data would produce the molarity if set above the volume. My data gave me the appropriate means (volume) to calculate this. The standardized permanganate was .061 M, which I put over 1L multiplied by the average volume of permanganate added, the mole to mole ratio, and volume of oxalic acid titrated, which resulted in .133 M. For this reason, I support my hypothesis.
There are many experimental errors that could have occurred throughout the experiment that would alter the results. To start, the iron used was purchased in 2012. Since the iron was the starting step, if it’s age caused it to be less reactive, if would affect the calculations throughout the whole experiment. Next, the true molarity of the iron was incorrect. The lab tech used MM instead of M. Without knowledge of this, all the calculations would be off because of decimal places. Once again, iron is the first titration, hindering the rest of the calculations from being correct. Last, if the stirring wasn’t consistent as the permanganate dropped into the iron or oxalic acid, then the exact point where the solution changed color may not have been noted correctly.
All of these experimental errors can easily be fixed to improve the results, and to make them more accurate. As for the age of the iron, simply but newer iron. And for the miscalculation of the M of the iron, be sure to double check everything as you work, or properly notify someone if a mistake has been made. That same rule applies for the stirring during a titration, be more cautious of what is going on. Further questions that came up due to this lab are as follows; what other acids or metals can be titrated by permanganate? How many titrations will we be doing this year? Are titrations always done the same way? How can you tell if a catalyst is needed to speed up the reaction?
Throughout the experiment, my hypothesis stood true. I started with moles of iron, followed all of the procedure, and did calculations to finish with the concentration of oxalic acid as said in my hypothesis. I stated that if we find the molarity of the potassium permanganate after it is standardized, then we can find the concentration of oxalic acid, because using a mole to mole conversion of the permanganate and the moles of oxalic acid found from the data would produce the molarity if set above the volume. My data gave me the appropriate means (volume) to calculate this. The standardized permanganate was .061 M, which I put over 1L multiplied by the average volume of permanganate added, the mole to mole ratio, and volume of oxalic acid titrated, which resulted in .133 M. For this reason, I support my hypothesis.
There are many experimental errors that could have occurred throughout the experiment that would alter the results. To start, the iron used was purchased in 2012. Since the iron was the starting step, if it’s age caused it to be less reactive, if would affect the calculations throughout the whole experiment. Next, the true molarity of the iron was incorrect. The lab tech used MM instead of M. Without knowledge of this, all the calculations would be off because of decimal places. Once again, iron is the first titration, hindering the rest of the calculations from being correct. Last, if the stirring wasn’t consistent as the permanganate dropped into the iron or oxalic acid, then the exact point where the solution changed color may not have been noted correctly.
All of these experimental errors can easily be fixed to improve the results, and to make them more accurate. As for the age of the iron, simply but newer iron. And for the miscalculation of the M of the iron, be sure to double check everything as you work, or properly notify someone if a mistake has been made. That same rule applies for the stirring during a titration, be more cautious of what is going on. Further questions that came up due to this lab are as follows; what other acids or metals can be titrated by permanganate? How many titrations will we be doing this year? Are titrations always done the same way? How can you tell if a catalyst is needed to speed up the reaction?
Discussion/ Reflection:
In this lab I learned a lot about oxidation-reduction reactions, half reactions, equivalence point, and titration in general. I also got more practice on stoichiometry and writing balanced net ionic equations. This relates to what we’re doing in class because many of the objectives were in this lab. For example, writing net ionic equations, balancing redox reactions, predicting products, identifying a substance as oxidized or reduced, and simply chemical reactions, which is the name of the unit. This applies to me in real life because I want to be an engineer when I grow up, and engineers use a lot of chemistry and chemical reactions to make things or make things better. In order to be an engineer I need to know these things, even if i’m not planning on being a chemical engineer. The lab experience was easier because we have been learning these concepts in class. The calculations would have been very difficult without prior knowledge and background on theses subjects.
In this lab I learned a lot about oxidation-reduction reactions, half reactions, equivalence point, and titration in general. I also got more practice on stoichiometry and writing balanced net ionic equations. This relates to what we’re doing in class because many of the objectives were in this lab. For example, writing net ionic equations, balancing redox reactions, predicting products, identifying a substance as oxidized or reduced, and simply chemical reactions, which is the name of the unit. This applies to me in real life because I want to be an engineer when I grow up, and engineers use a lot of chemistry and chemical reactions to make things or make things better. In order to be an engineer I need to know these things, even if i’m not planning on being a chemical engineer. The lab experience was easier because we have been learning these concepts in class. The calculations would have been very difficult without prior knowledge and background on theses subjects.