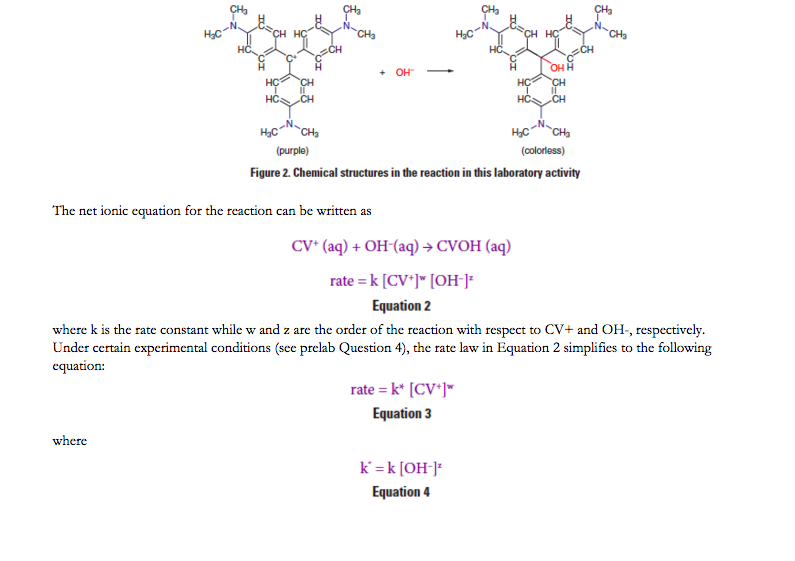
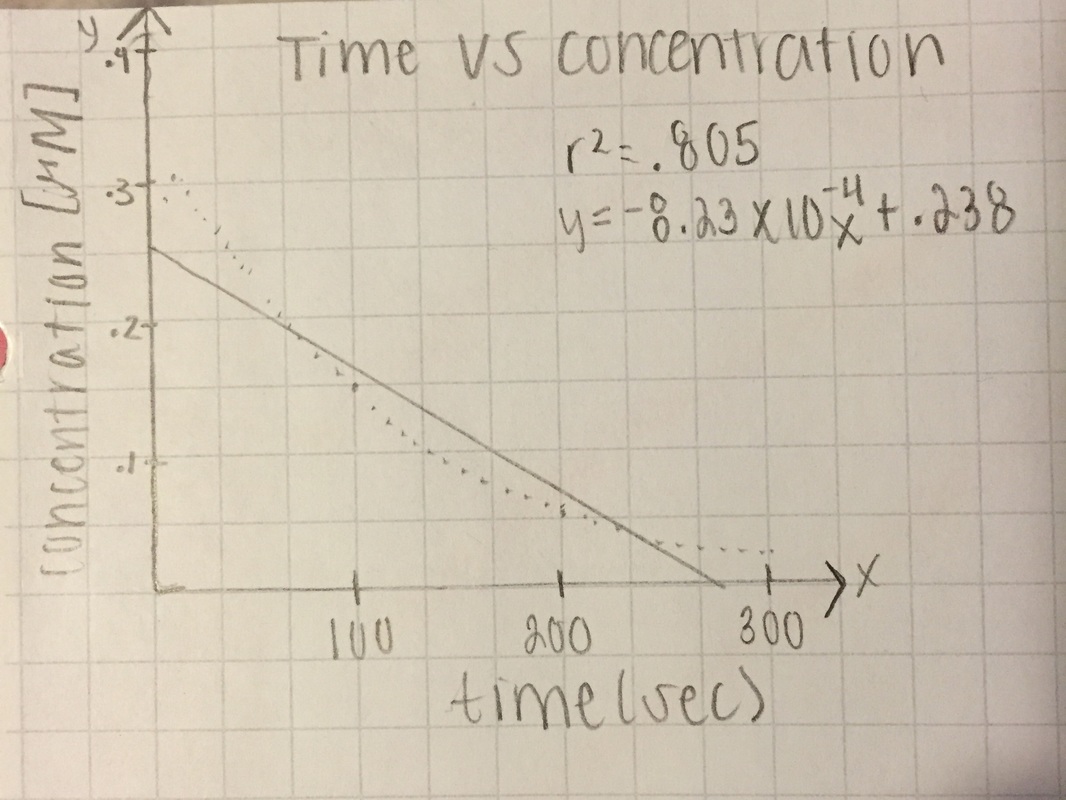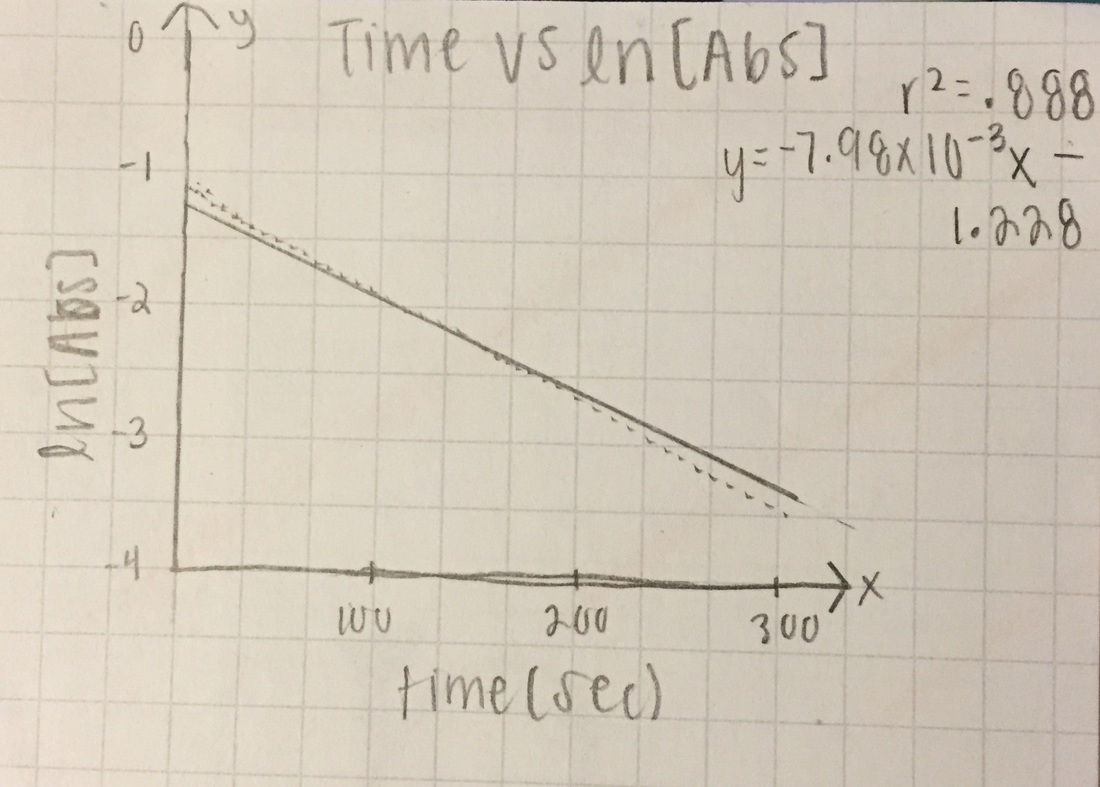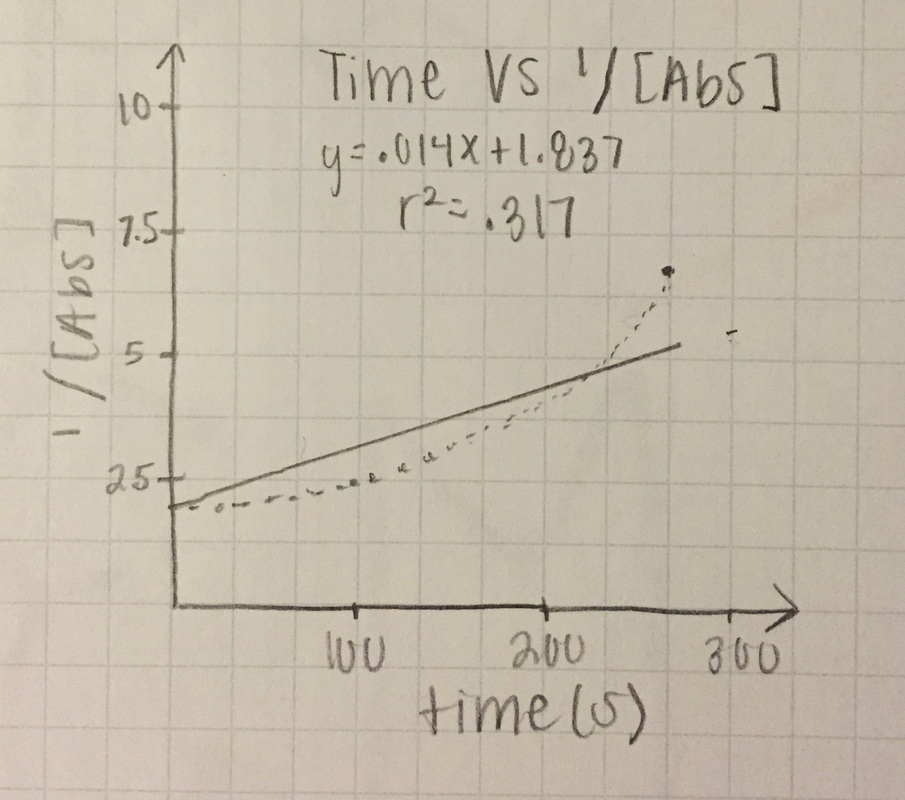Crystal Violet Rate Law Lab
January 27, 2016 AP Chemistry pd.4
Rileigh Robertson
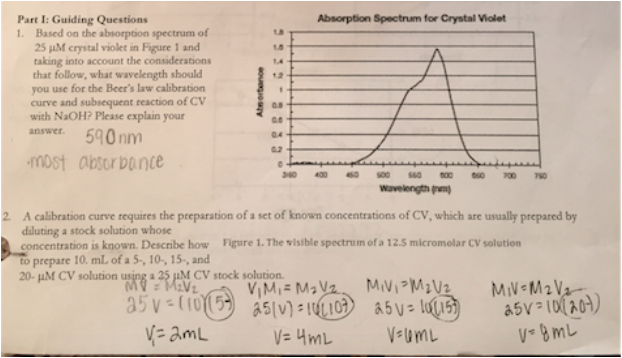
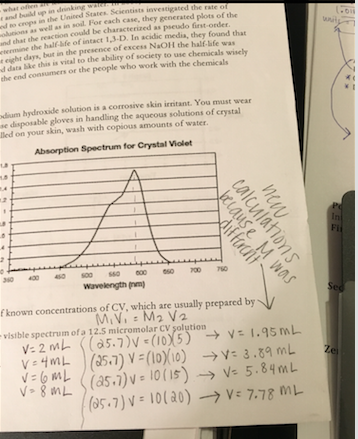
The lab originally said to dilute a 25 micro-molar solution as the "pre-lab" but the solution we got was 25.7 micro molar, so I redid the calculations. (below)
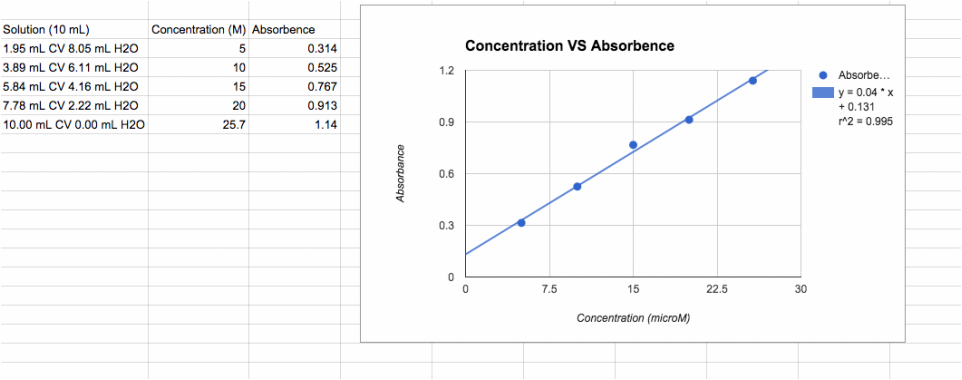
5. Calibration curve for dilutions.
Analysis:
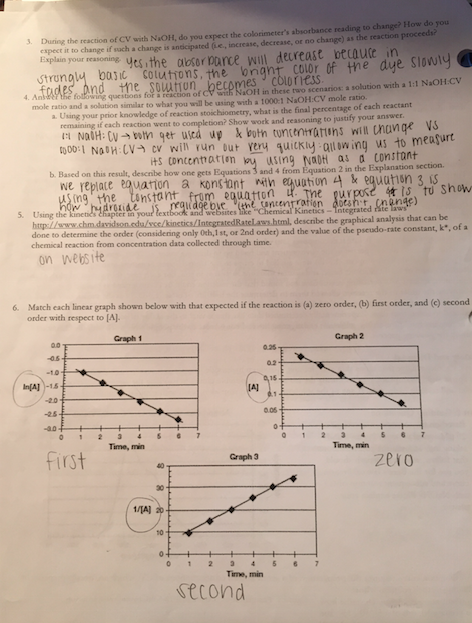
**Something to note: Absorption and concentration are directly related (nearly the same) so I just used Absorbance for my data!
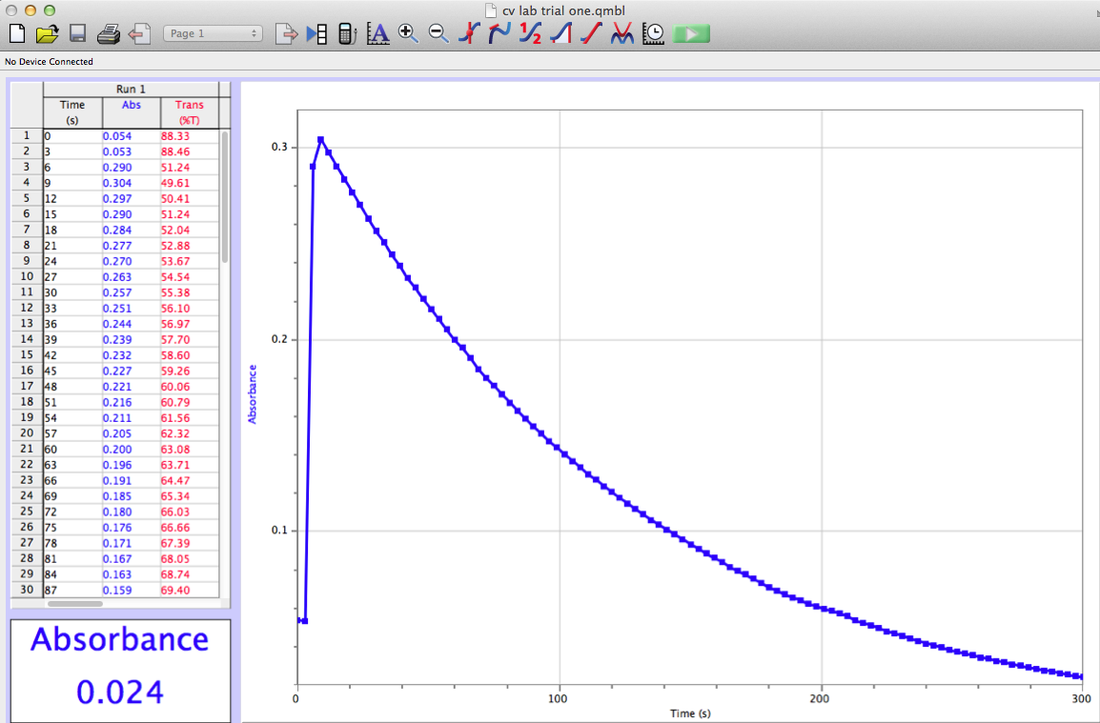
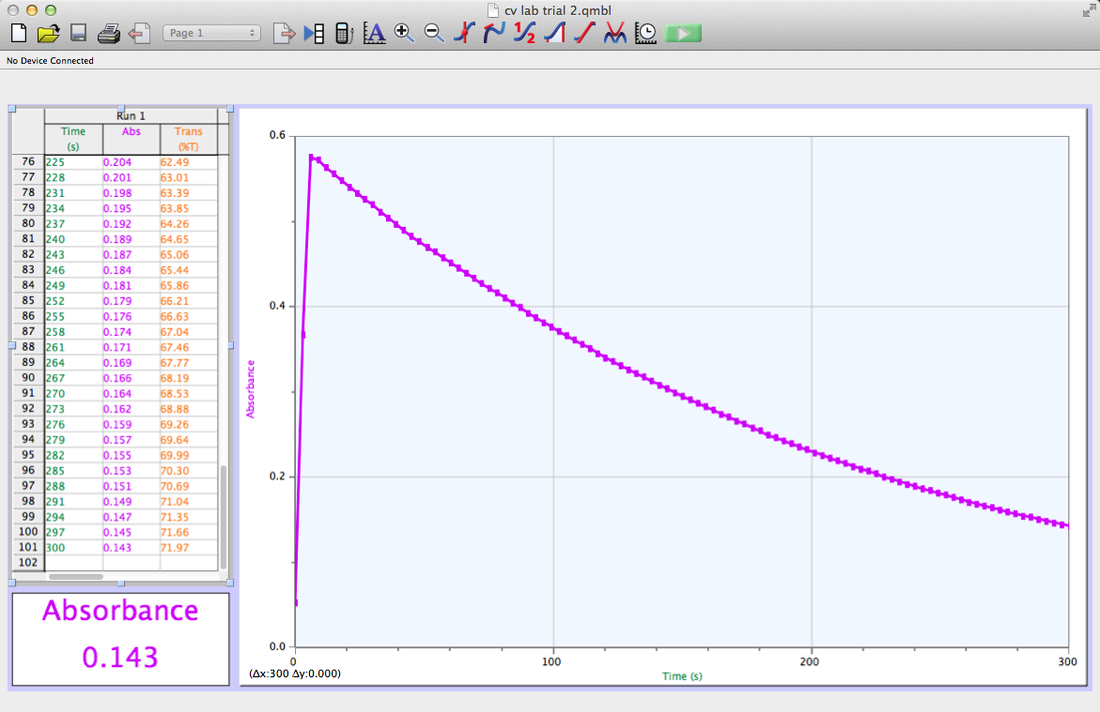
Trial 1
I did my graphs on excel instead of my graphing calculator because there were so many points, and I wanted to have them all to make my data more accurate.
1-4. Shown Above
5.
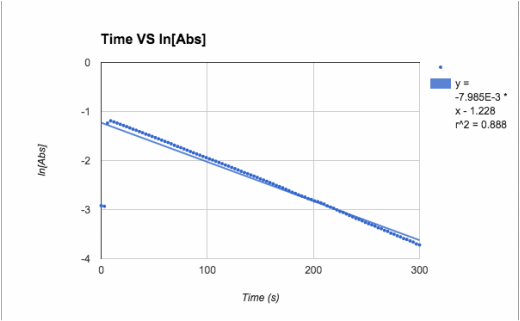
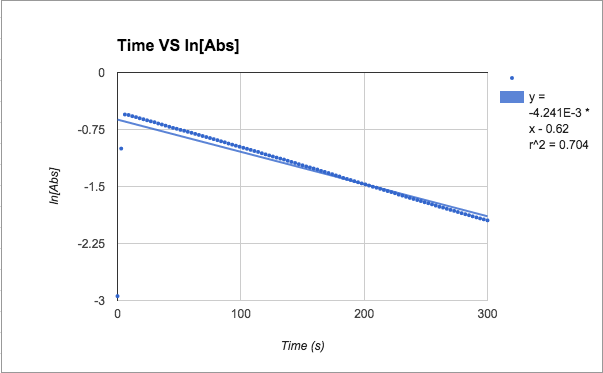
1st order: ln[Abs]
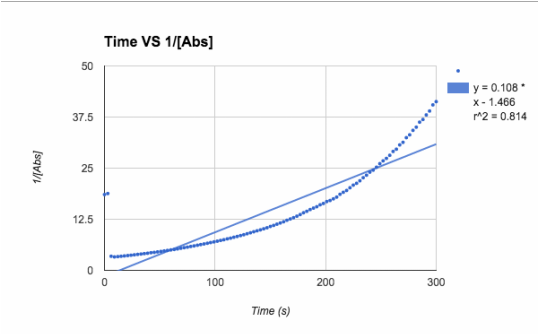
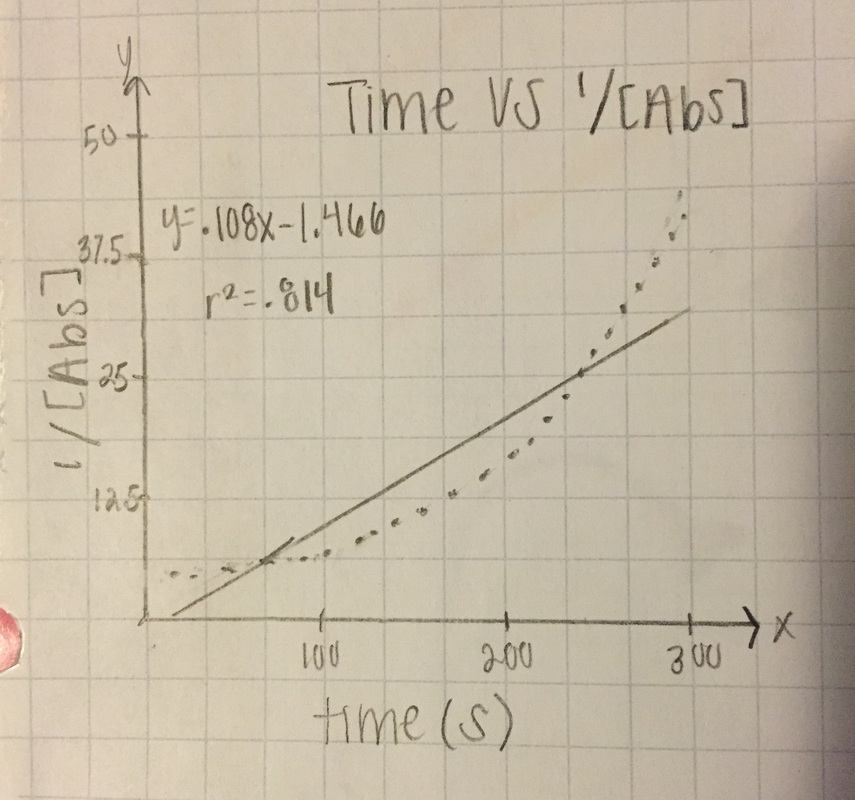
2nd order: 1/[Abs]
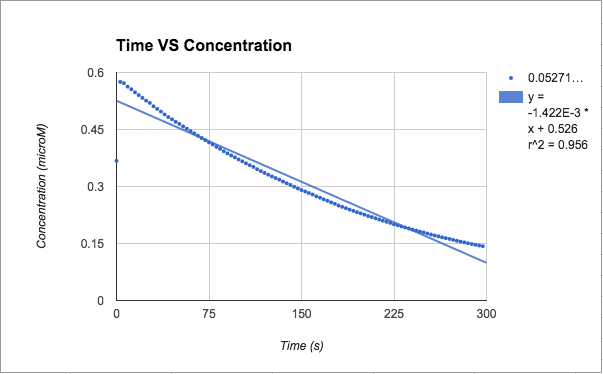
Zero order: [A] just concentration
1-4. Shown Above
5.
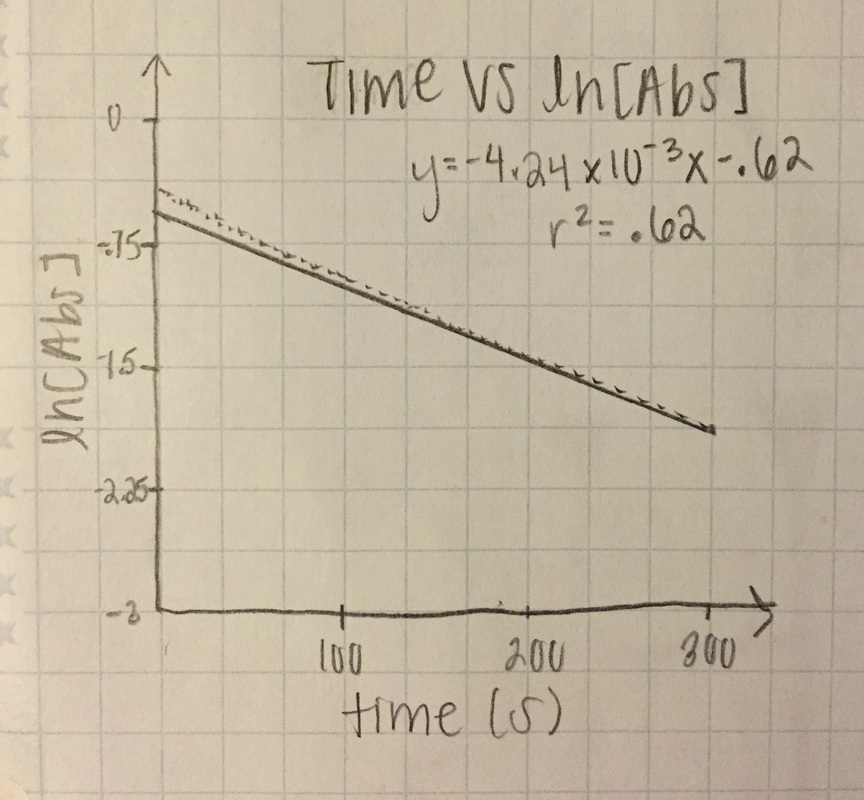
1st order: ln[Abs]
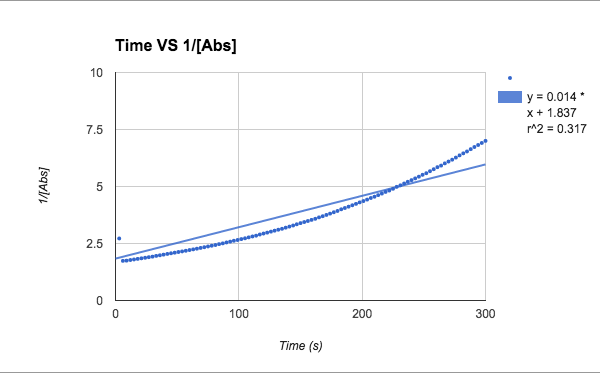
2nd order: 1/[Abs]
Zero order: [A] just concentration
Trial 2
I did my graphs on excel instead of my graphing calculator because there were so many points, and I wanted to have them all to make my data more accurate.
1-4. Shown Above
5.
1st order: ln[Abs]
2nd order: 1/[Abs]
Zero order: [A] just concentration
6.
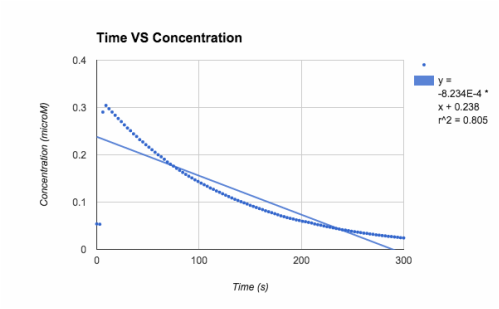
[A] - both slopes are in negative, very small, values. trial 2 has the r^2 value closest to one (.956), but trial 1 was till decent at .805
T= trial slope T1/T2= -8.23x10^-4/-1.422x10^-3 = 57.9% accurate
ln[A] - both slopes are in negative, very small (x10-3), values. Trial 1 has the better r^2 value at .888 while trial 2 really dropped (.62)
T= trial slope T2/T1 (since T2 is bigger)= -4.24x10^-3/-7.98x10^-3 = 53.1% accurate
1/[A] - both slopes are in the positive zeros, very small numbers, and trial 1 keeps is consistent with an R62 value of .814, and trial 2 r^2 value drops even more to .317
T= trial slope T2/T1 (since T2 is bigger)= .014/.108 = 13.0% accurate
7.
Overall Rate Law:
rate=k[CV]^1[OH]^-.785
according to a slope to OH ratio
1-4. Shown Above
5.
1st order: ln[Abs]
2nd order: 1/[Abs]
Zero order: [A] just concentration
6.
[A] - both slopes are in negative, very small, values. trial 2 has the r^2 value closest to one (.956), but trial 1 was till decent at .805
T= trial slope T1/T2= -8.23x10^-4/-1.422x10^-3 = 57.9% accurate
ln[A] - both slopes are in negative, very small (x10-3), values. Trial 1 has the better r^2 value at .888 while trial 2 really dropped (.62)
T= trial slope T2/T1 (since T2 is bigger)= -4.24x10^-3/-7.98x10^-3 = 53.1% accurate
1/[A] - both slopes are in the positive zeros, very small numbers, and trial 1 keeps is consistent with an R62 value of .814, and trial 2 r^2 value drops even more to .317
T= trial slope T2/T1 (since T2 is bigger)= .014/.108 = 13.0% accurate
7.
Overall Rate Law:
rate=k[CV]^1[OH]^-.785
according to a slope to OH ratio