Atomic Emission Spectra Lab Report (Abstract)
Rileigh Robertson pd.4 11/20/15
Pre-Lab Questions
Purpose
The purpose of this experiment was to observe the emission spectrum of atoms in a flame test.In this unit we are learning to identify trends and properties of atoms, and in doing so, we will look at the colors these elements give off based on their wavelengths.
Procedure
In this experiment, we tested the wavelengths of Li, Sr, K, Mg, Ba, Cu, and Ca by holding a strip of each of these metals over an open flame, and observed the color of light that the the metal gave off through a spectroscope. The spectroscope gave us a value which we later used to find the wavelengths.
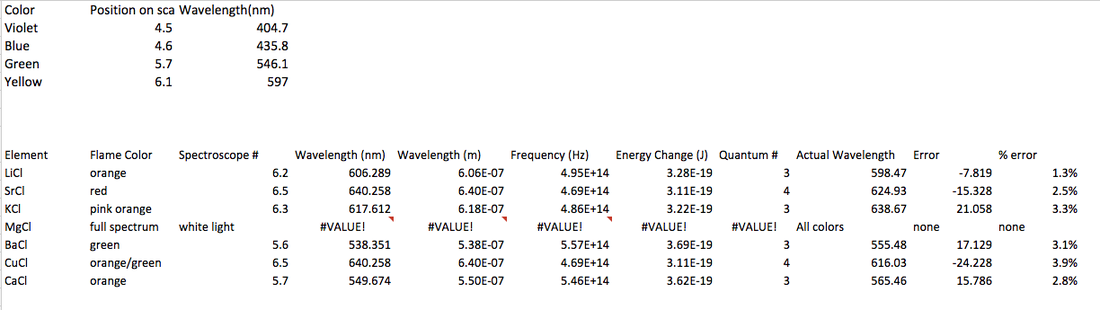
Observations/ Data/ Results
We found lithium to be orange (6.2), strontium to be red (6.5), potassium to be pink/orange (6.3), magnesium to be white light (full spectrum), barium to be green (5.6), copper to be orange/green (6.5), and calcium to be orange (5.7). From this we found out about how the electrons effect the light given off.
Conclusions
I believe that as you increase the group number on the periodic table, the element becomes less reactive with a larger wavelength, hence the color change. For example, strontium was a bright red color with a large wavelength, and a smaller frequency than the rest. This pattern is also true for the energy of the elements.
This experiment shows how wavelength directly relates to the frequency and energy all derived from the visible light spectrum. As for real world applications, several different professions would find this useful including engineering. Also, you see color all day everyday, and know you will know how it comes from electron behavior.
This experiment shows how wavelength directly relates to the frequency and energy all derived from the visible light spectrum. As for real world applications, several different professions would find this useful including engineering. Also, you see color all day everyday, and know you will know how it comes from electron behavior.
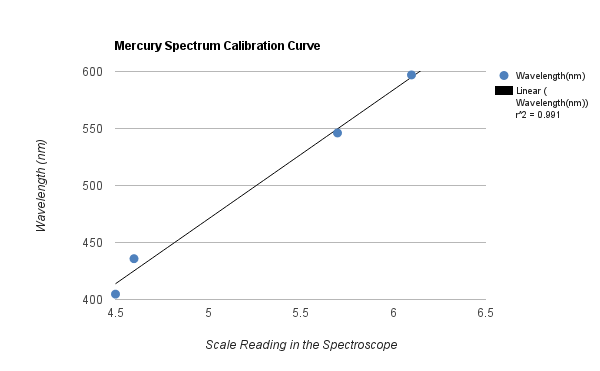
Data Analysis
1. y=113.23x-95.737
r value= .99134
r value= .99134
2 and 9. Our class did not have time to do th emission spectrum for hydrogen or test the unknowns due to a snow day.
3-8. below
3-8. below